New Medication Choice

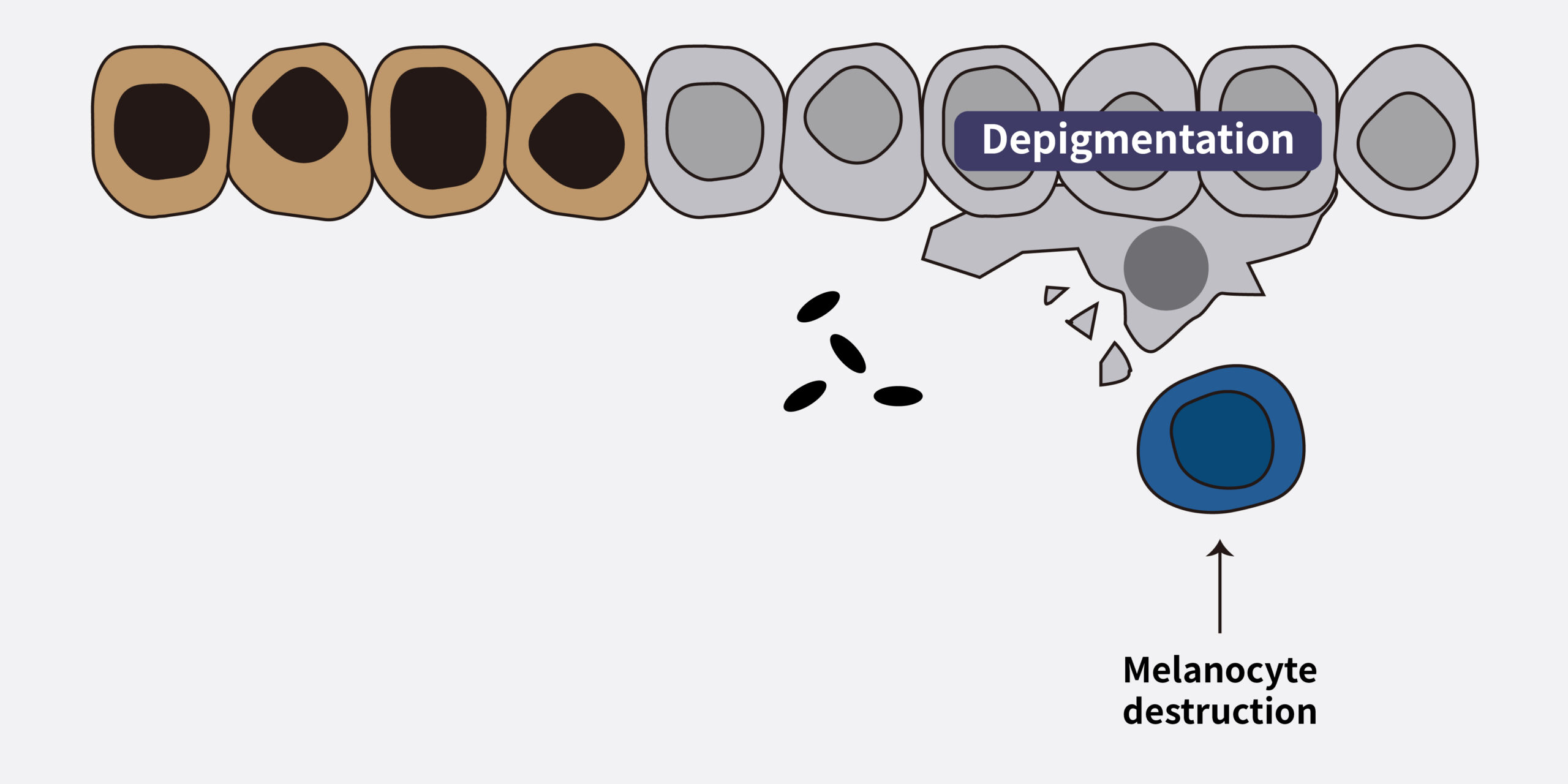
The goals of vitiligo treatment include stabilizing the condition, achieving repigmentation, and preventing recurrence. Recently, topical JAK inhibitors have been explored as a promising therapeutic option for vitiligo. The activation of the JAK-STAT pathway and the inflammatory cycle led to the destruction of melanocytes, resulting in depigmentation.3
Topical JAK inhibitors block the JAK-STAT pathway, which reduces JAK-STAT signaling and suppresses inflammatory responses. This helps to prevent the body’s attack on melanin-producing cells. Therefore, the JAK-STAT pathway presents a compelling target for therapeutic intervention. 3,4
Clinical studies on the effectiveness of topical JAK inhibitor have shown promising results. In clinical trials, the application of a JAK inhibitor cream resulted in greater repigmentation in non-segmental vitiligo compared to the control group over a 52-week period.*5
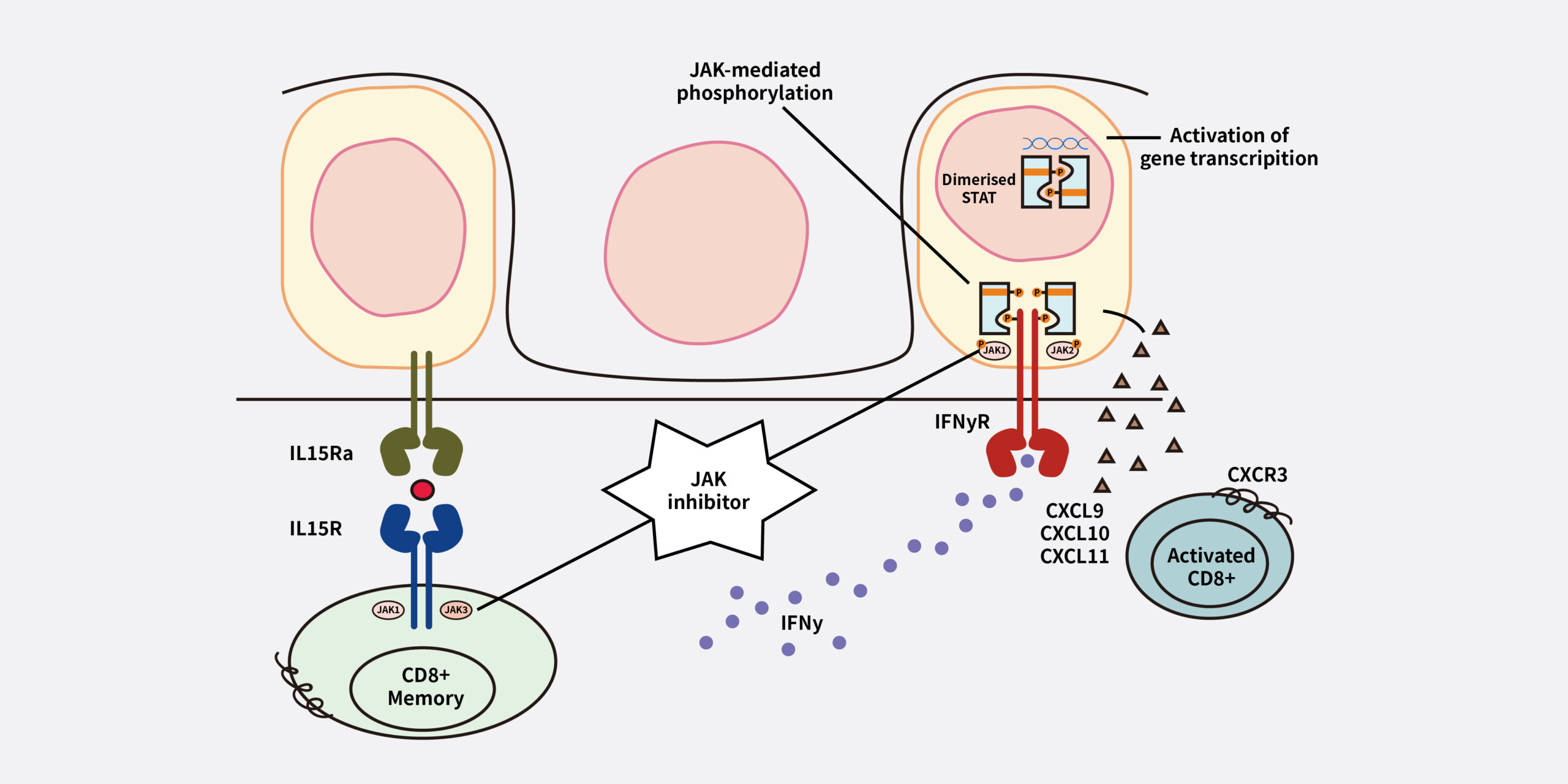
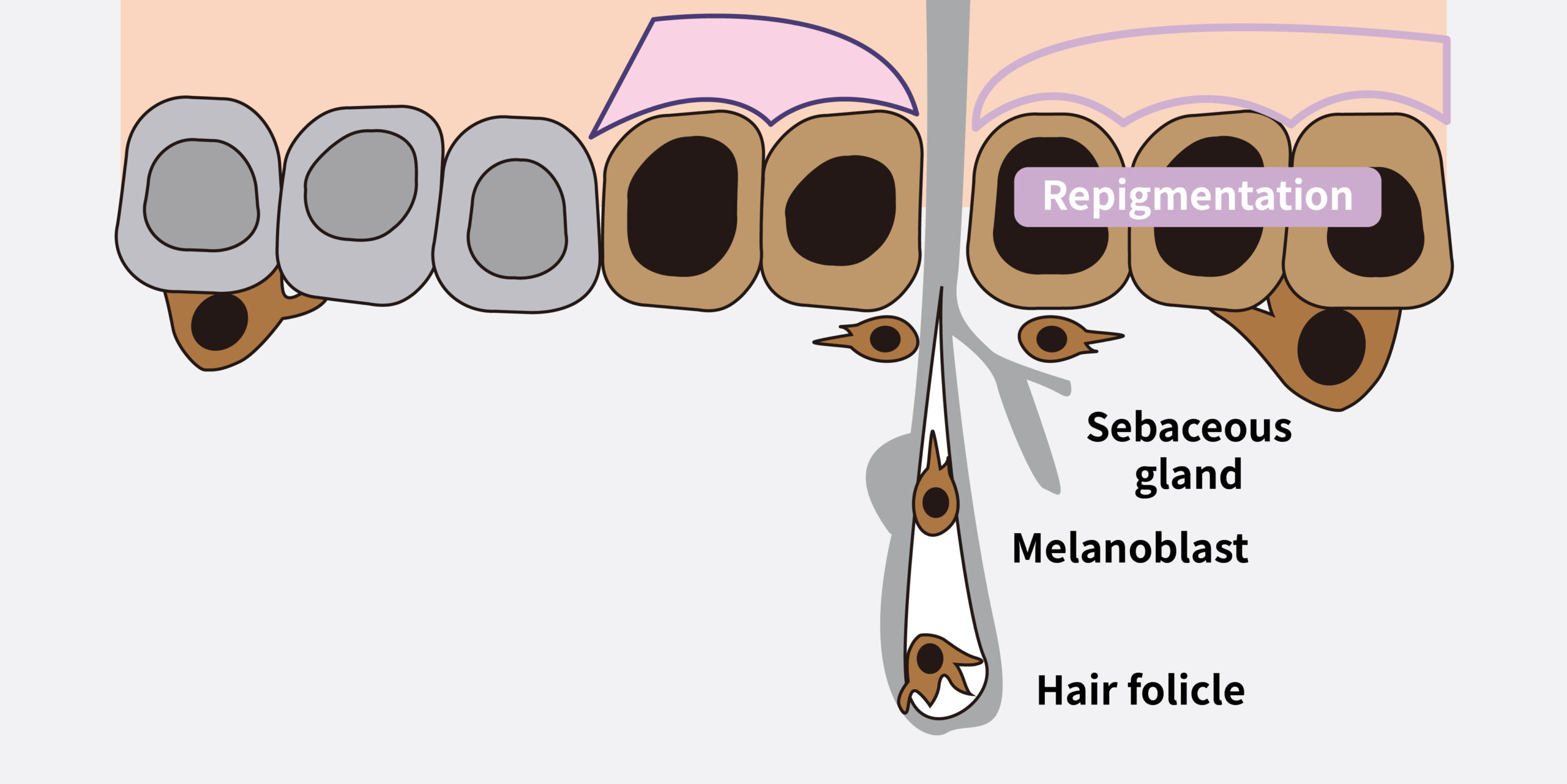
Pathophysiology of vitiligo and role of JAK inhibitors3
(Adapted from Utama A 2024)